Potent Compound Suite for Small Molecule Oncologicals

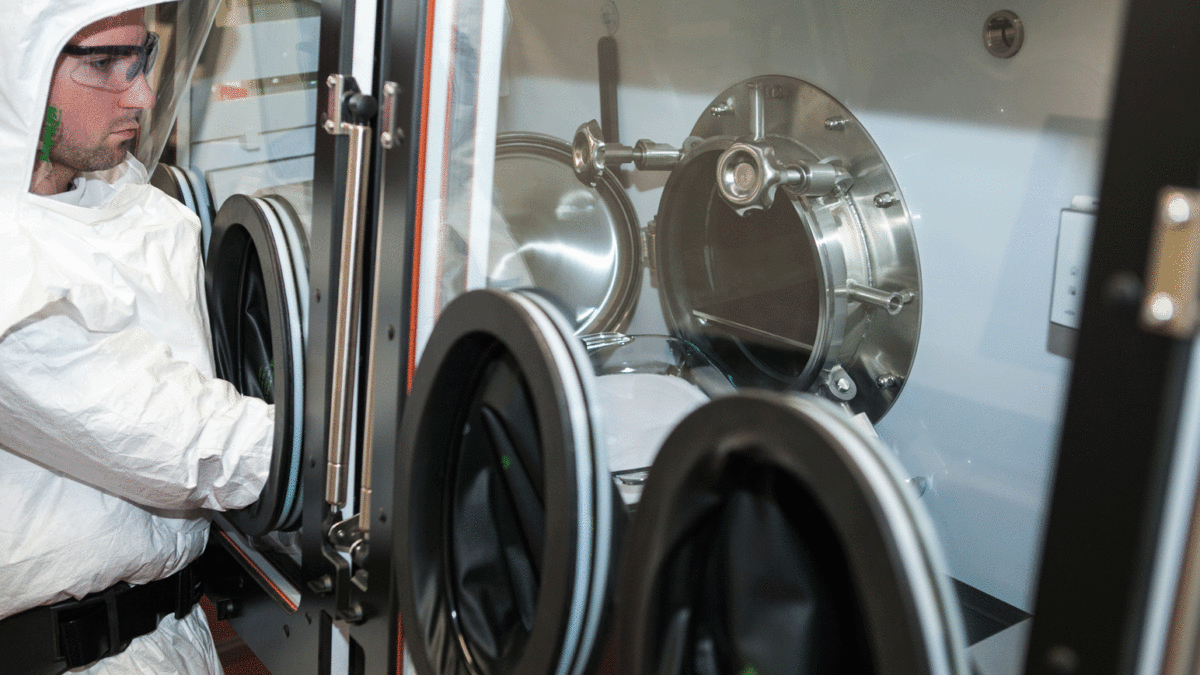
Regis Technologies is announcing their entry into the oncology market with the start up of the new Potent Compound Suite (PCS). The PCS allows Regis, a Chicago-based contract manufacturing organization (CMO), to target small molecule oncologicals and linker payload projects for antibody drug conjugates.
Regis is a CMO operating a 36,000 square foot, cGMP facility in Morton Grove, Illinois. The PCS addition provides for small molecule cGMP manufacturing of potent compounds up to about one kilogram per batch.
Regis’ Director of Business Development states “We see the PCS as an important asset to serve the growing oncology market as more companies look to advance their lead candidates into the clinic.”
Regis’ Manager of Process Engineering, Dragan Ristic, supervised the design and construction of the suite. Proper handling of potent compounds requires the use of specialized facilities that are carefully designed to minimize containment risks. Regis’ new PCS includes isolators, laminar flow hoods and local exhaust ventilation appropriate for potent compound handling.
Regis worked with high containment industry consultants to choose the best laboratory and personal protective equipment for potent compound handling and conducted employee training. Regis also completed validations and cGMP qualifications for the suite.
In 2015, the PCS passed the commissioning process. Safebridge Consultants, Inc. evaluated the containment performance of the facility via industrial hygiene monitoring. Based on the occupational exposure limits (OELs) of the future Active Pharmaceutical Ingredients (APIs) to be handled, a containment performance target (CPT) was set to 50 ng/m3.
The surrogate monitoring involved handling of surrogate material (naproxen sodium) in PCS by Regis’ employees, while performing the air and surface sampling by certified industrial hygienist in order to find out how much of material could escape from the containment. Read more about the process here.
The report described the testing methodology and provided an objective interpretation of the results indicating that the containment performance of the equipment is in full compliance with the Regis’ CPT. Personal and area measurements for surrogate powder handling showed that containment isolator system in the PCS1 can be used to handle a wide range of potent APIs.
The surface sampling results were found to be well below the performance-based acceptable surface limit (ASL) for SafeBridge Category 3 (Regis HHC 4) compounds. This evaluation gives further confidence that potent APIs are being handled safely at Regis Technologies, protecting employees, products, and environment.
Regis is excited to add the capabilities of the PCS to our 60 year long resume of supporting biotech and pharma customers. Founded in 1956, Regis has long been recognized as a premier partner for the clinical and commercial production of small molecule APIs. Its outstanding compliance history, technical and project management teams, and central US location have made it the supplier of choice for emerging and mid-size pharma.
For more information on highly potent active pharmaceutical manufacturing and additional pharma services, click here.